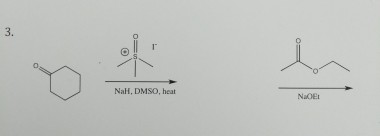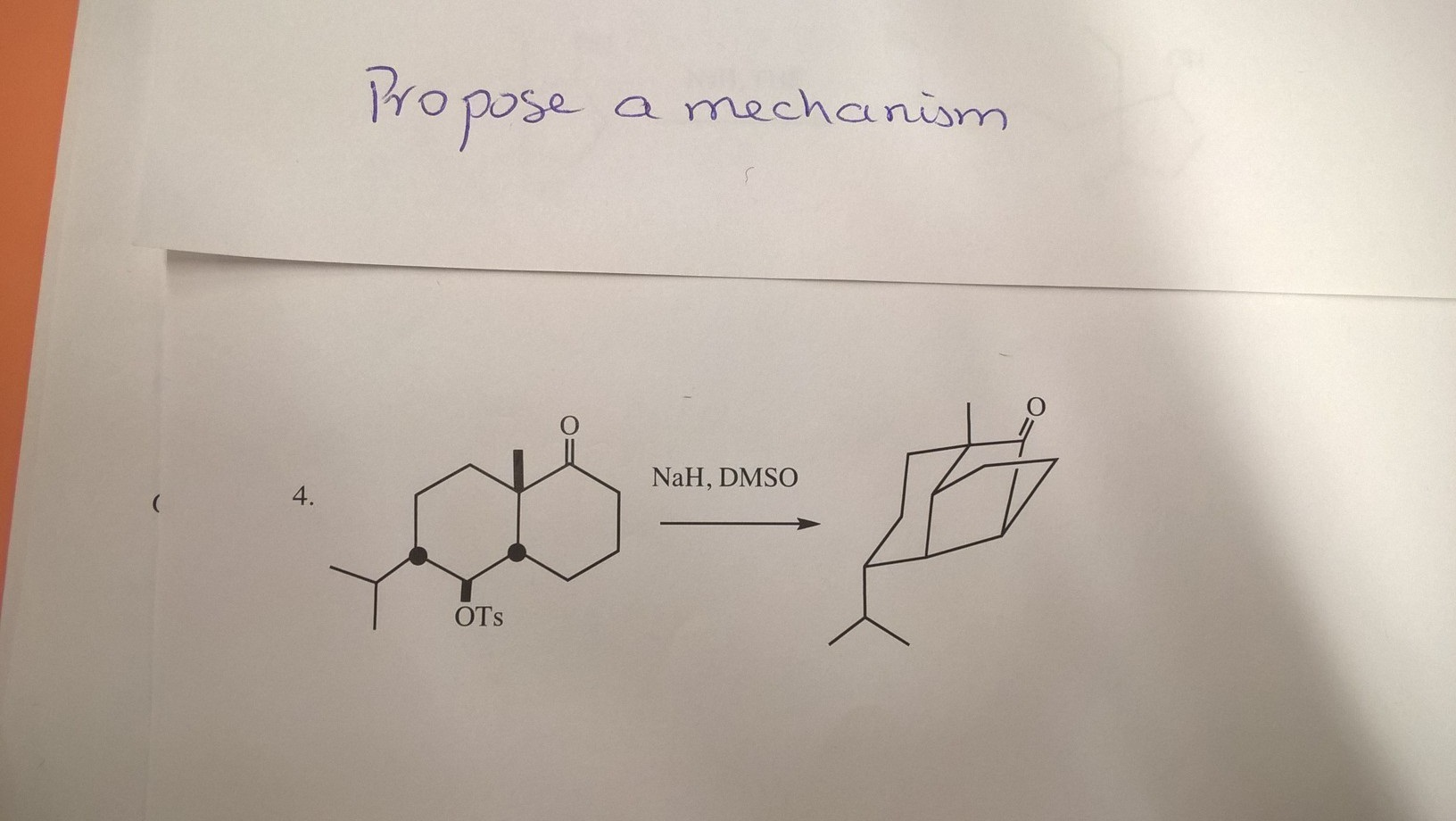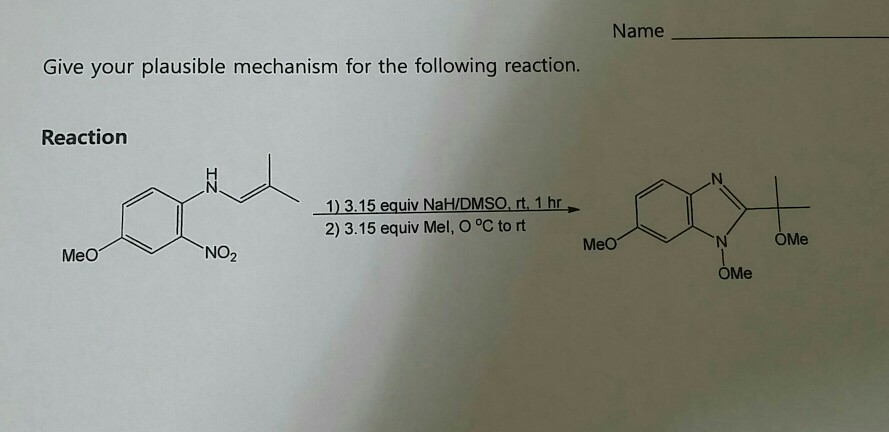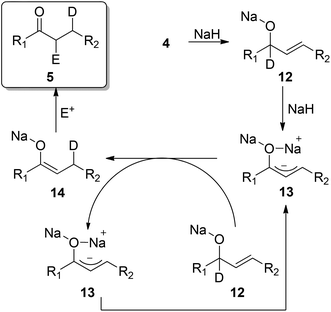
NaH mediated isomerisation–allylation reaction of 1,3-substituted propenols - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C3OB41857J
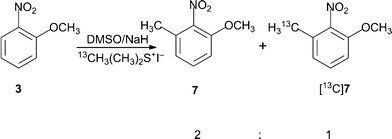
Concerning the deprotonation of the trimethylsulfonium ion by the dimethylsulfinyl anion - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C1OB05889D

Synthetic pathway for compounds (42-49). Reagents and conditions: (a)... | Download Scientific Diagram
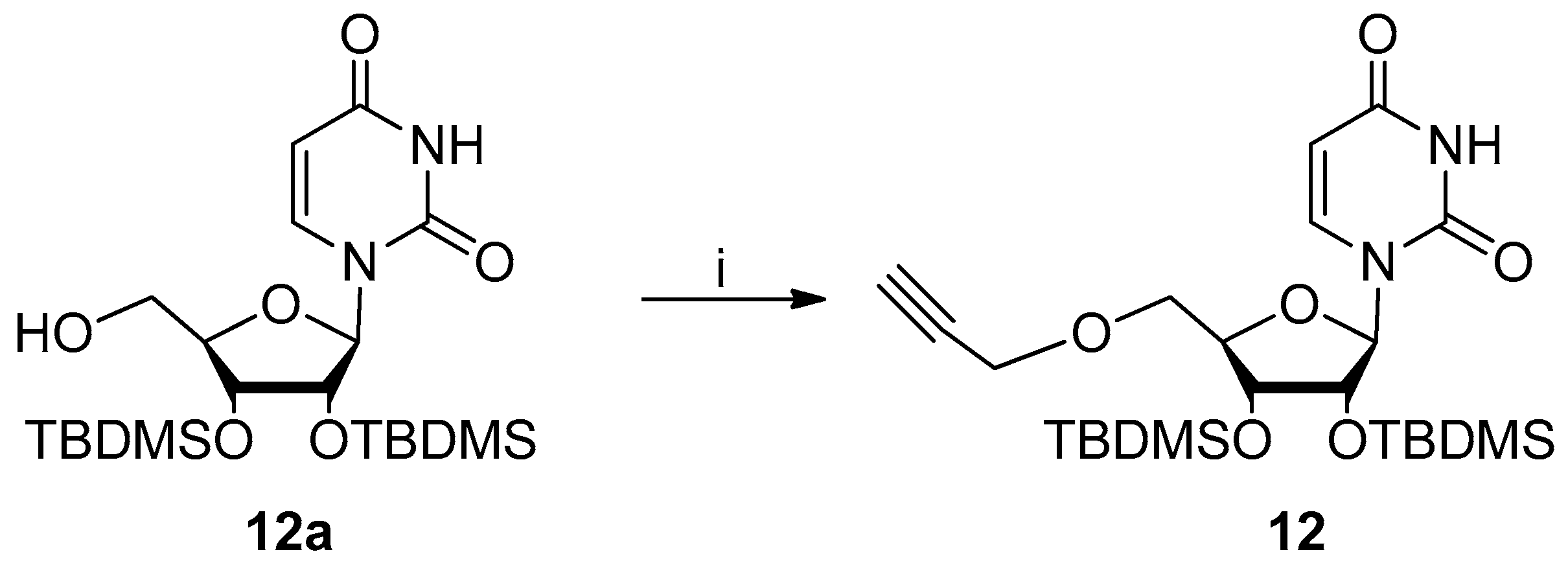
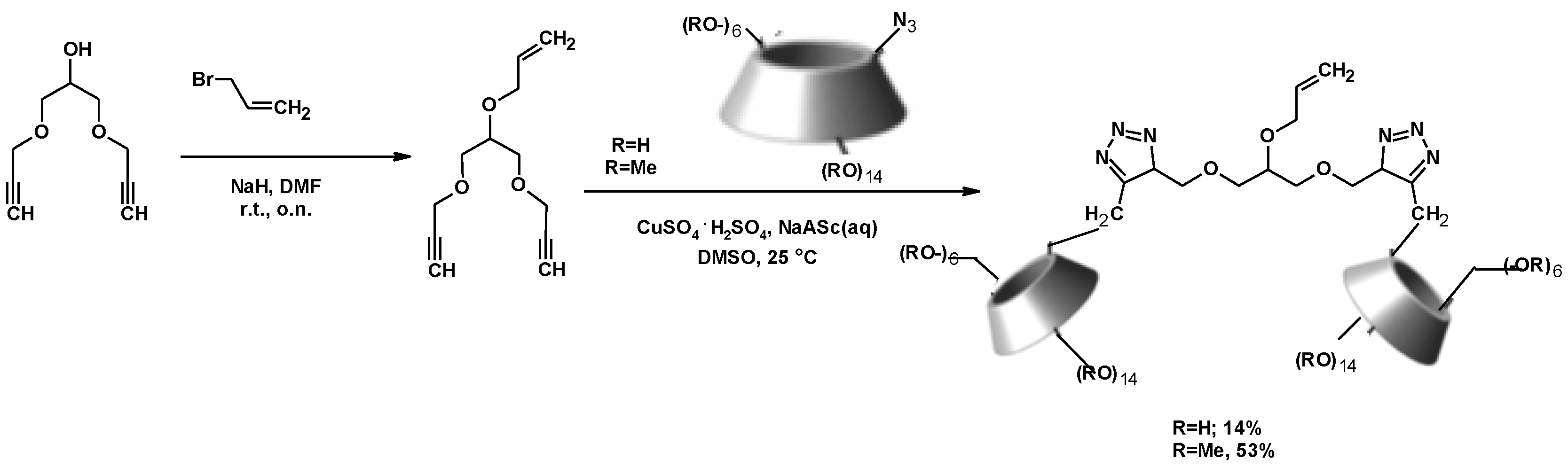
Pharmaceuticals | Free Full-Text | Anti-Tick-Borne Encephalitis Virus Activity of Novel Uridine Glycoconjugates Containing Amide or/and 1,2,3-Triazole Moiety in the Linker Structure | HTML

Scheme 35 Reagents and conditions: (a) TNSI, NaH, DMSO, 86%; (b) 781,... | Download Scientific Diagram

Scheme 2. Reagents and Conditions: (i) NaH, Me 3 SI, DMSO, rt, 2 h,... | Download Scientific Diagram

Explosion Hazards of Sodium Hydride in Dimethyl Sulfoxide, N,N-Dimethylformamide, and N,N-Dimethylacetamide | Organic Process Research & Development

The Rich Chemistry Resulting from the 1,3‐Dipolar Cycloaddition Reactions of Enamines and Azides - Bakulev - 2018 - European Journal of Organic Chemistry - Wiley Online Library
![Organics | Free Full-Text | A Review of the Synthetic Strategies toward Dihydropyrrolo[1,2-a]Pyrazinones | HTML Organics | Free Full-Text | A Review of the Synthetic Strategies toward Dihydropyrrolo[1,2-a]Pyrazinones | HTML](https://www.mdpi.com/organics/organics-02-00011/article_deploy/html/images/organics-02-00011-sch010.png)
Organics | Free Full-Text | A Review of the Synthetic Strategies toward Dihydropyrrolo[1,2-a]Pyrazinones | HTML

Explosion Hazards of Sodium Hydride in Dimethyl Sulfoxide, N,N-Dimethylformamide, and N,N-Dimethylacetamide | Organic Process Research & Development

Applications of Continuous-Flow Photochemistry in Organic Synthesis, Material Science, and Water Treatment | Chemical Reviews

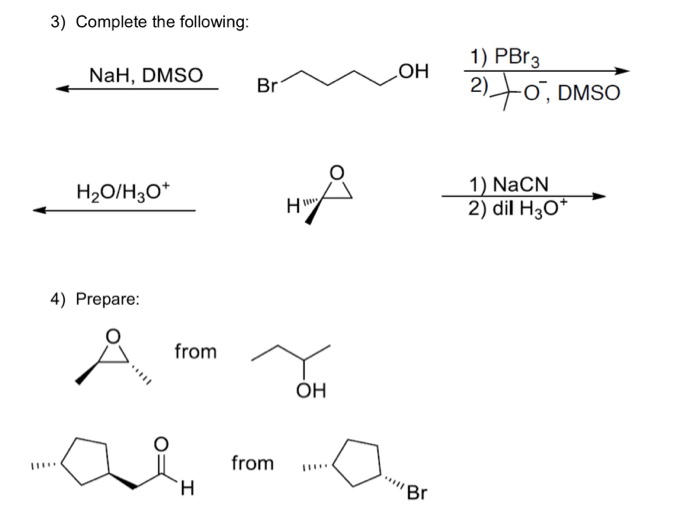
![Solved 4. [4 pts] HOCH2CH2Br reacts with base to form a | Chegg.com Solved 4. [4 pts] HOCH2CH2Br reacts with base to form a | Chegg.com](https://media.cheggcdn.com/media%2F1ae%2F1ae8b786-ce20-4a4d-ad01-aa7be83da65f%2Fimage)