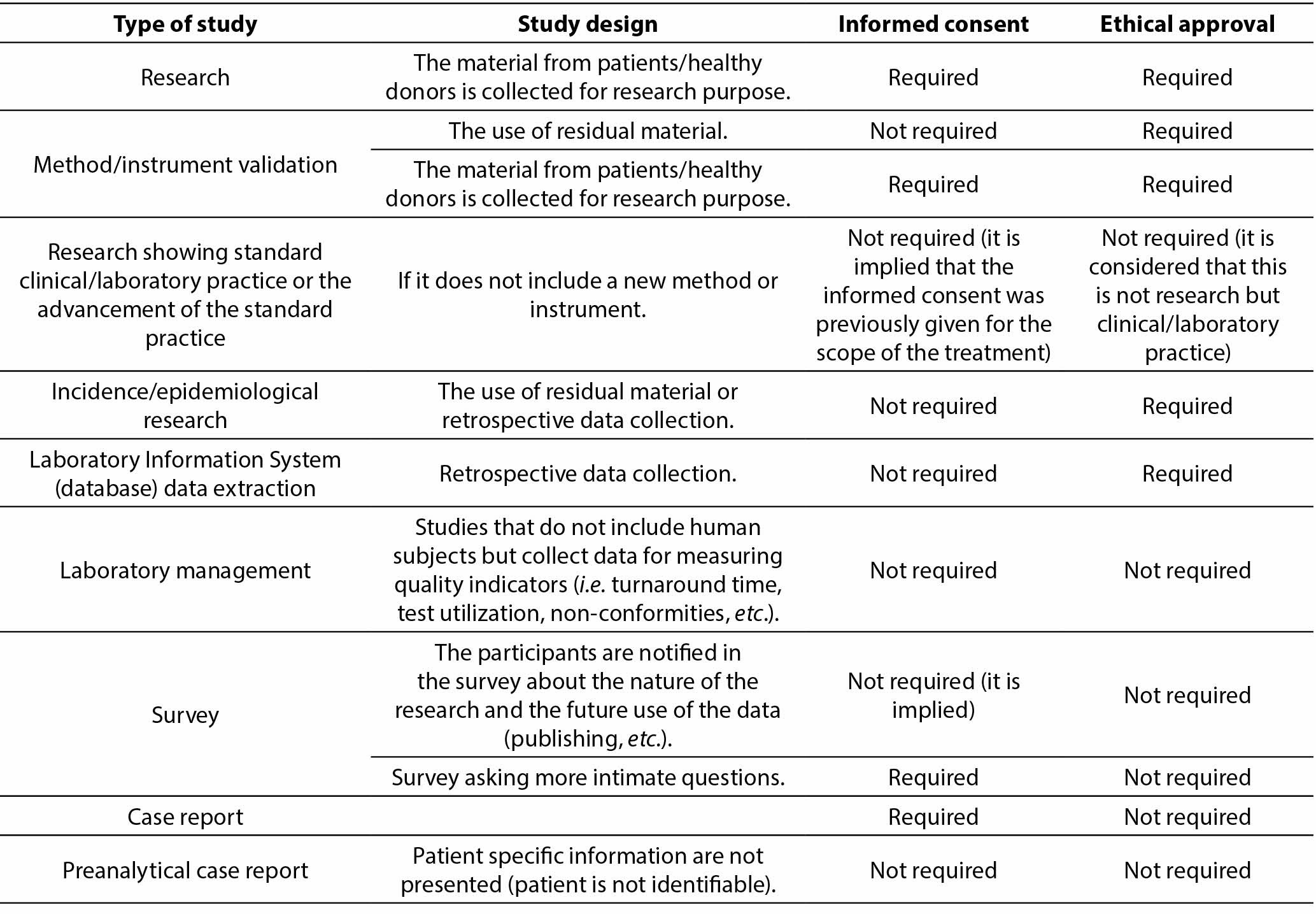
The Patient Information Sheet (PIS) and Informed Consent (IC) for case reports and case series: Proposal for a standard model for presentations in congresses and other scientific publications - ScienceDirect

Informed consent formulier - ............ Ik heb de informatiebrief gelezen en ben op deze manier - StudeerSnel