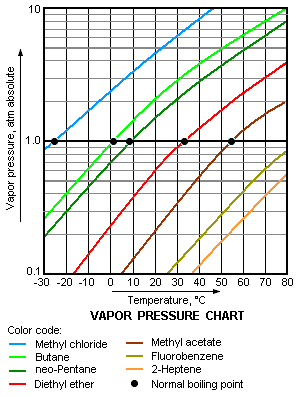
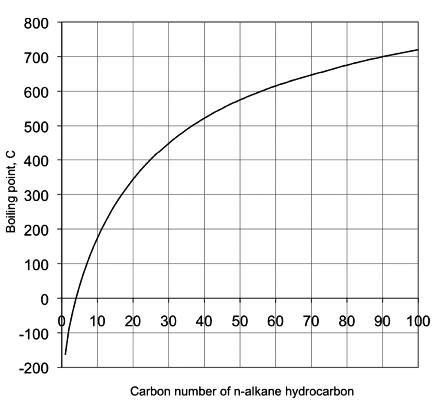
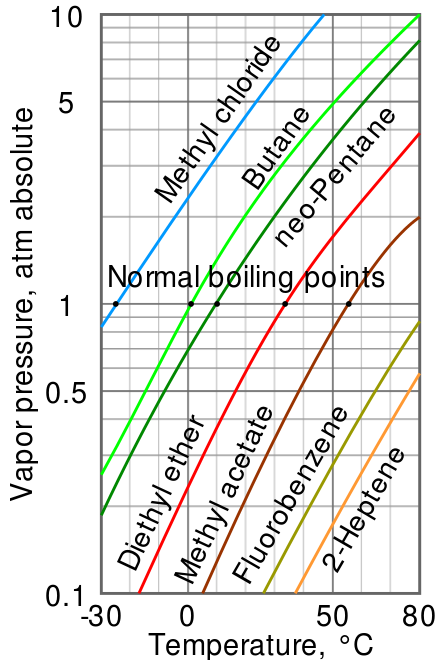
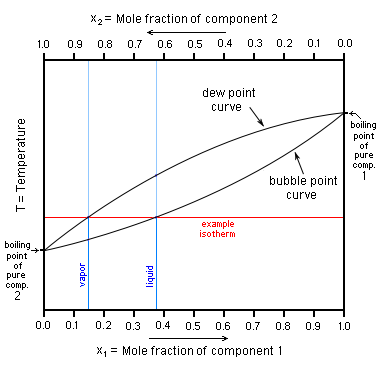
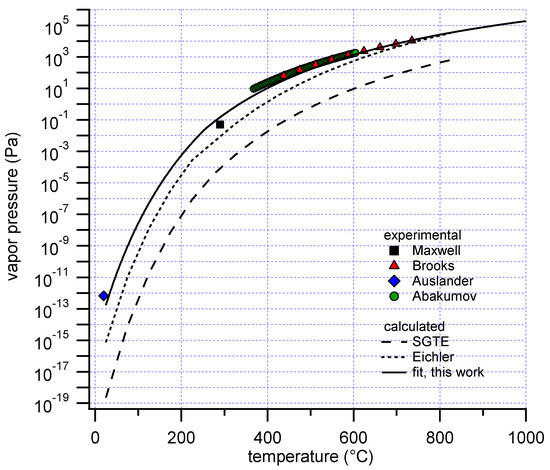
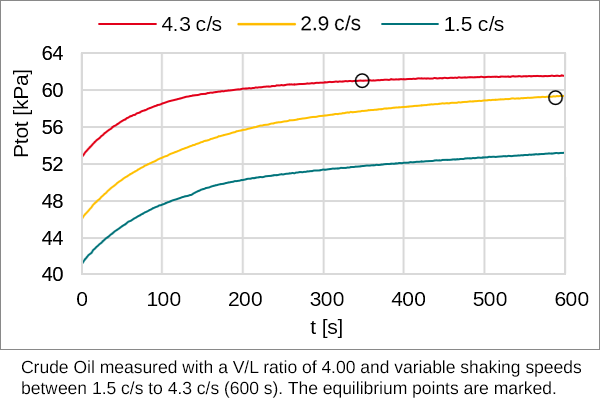
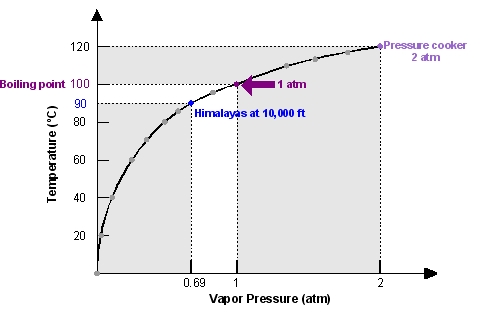
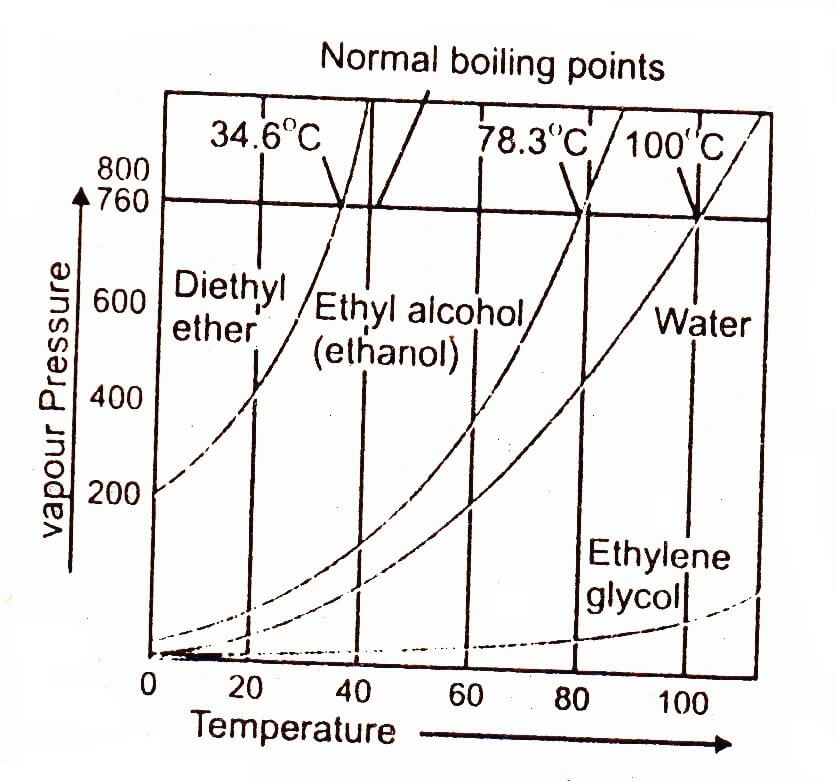
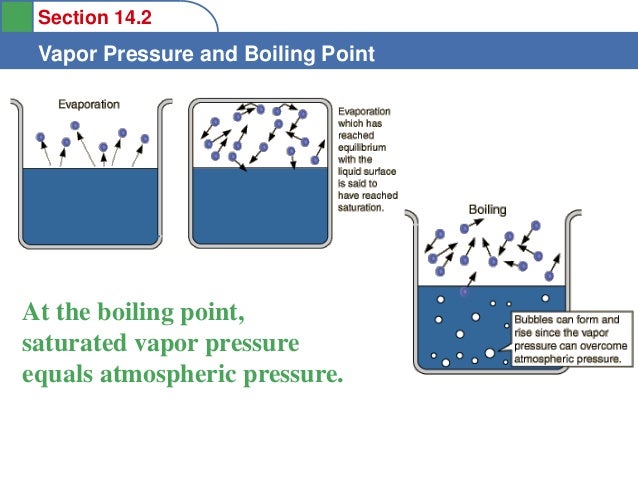
Diagram P-T relationship with the vapor pressure (A) solution boiling... | Download Scientific Diagram
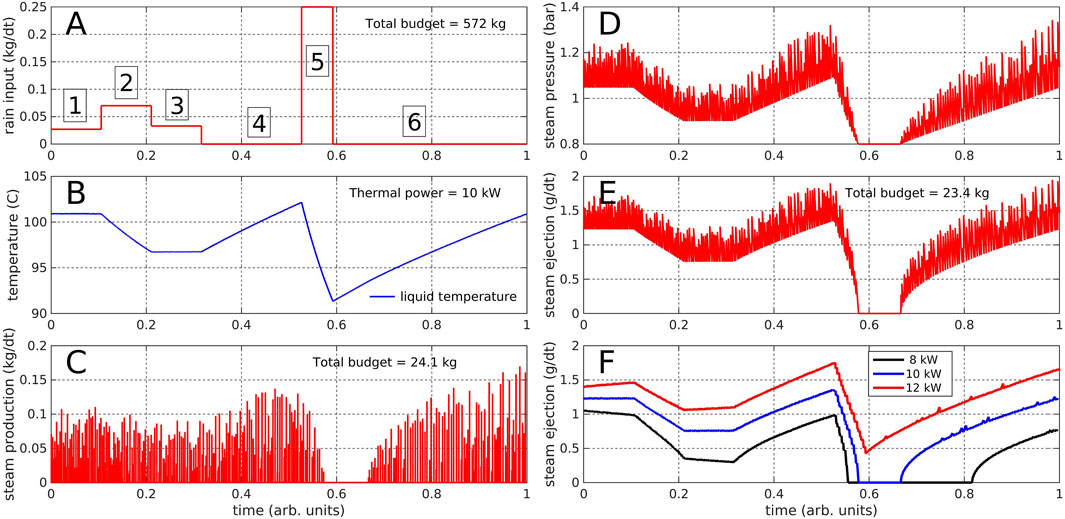
Frontiers | Low-Temperature Hydrothermal Systems Response to Rainfall Forcing: An Example From Temperature Time Series of Fumaroles at La Soufrière de Guadeloupe Volcano | Earth Science