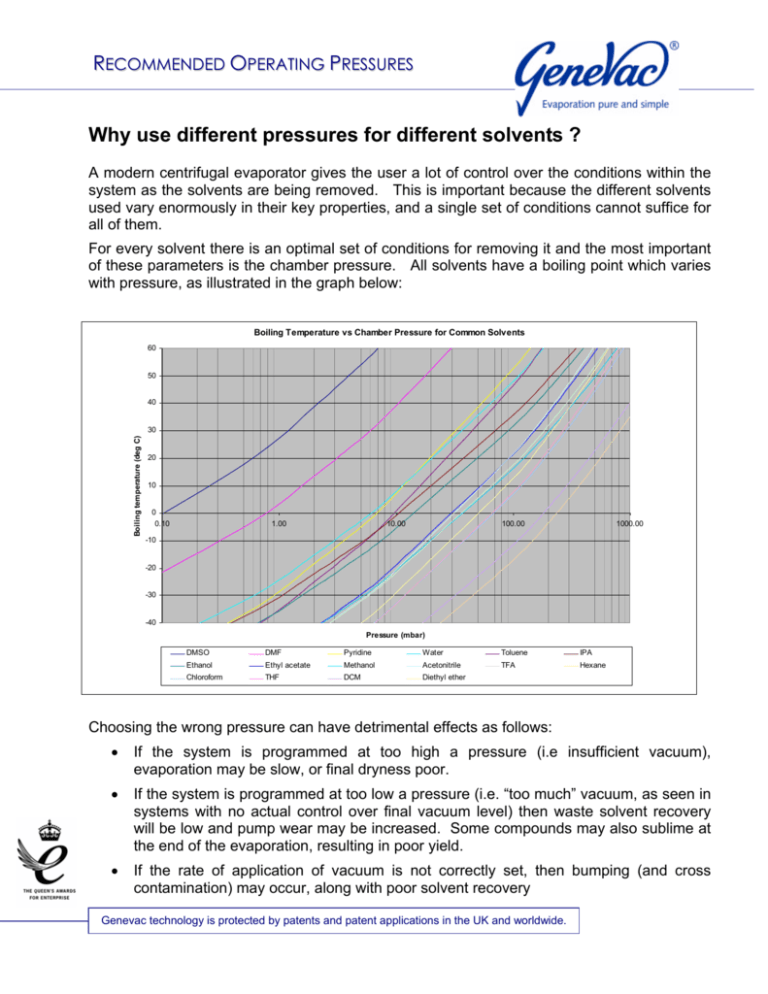
In Vitro Human Skin Absorption of Solvent-deposited Solids: Niacinamide and Methyl Nicotinate - Journal of Pharmaceutical Sciences
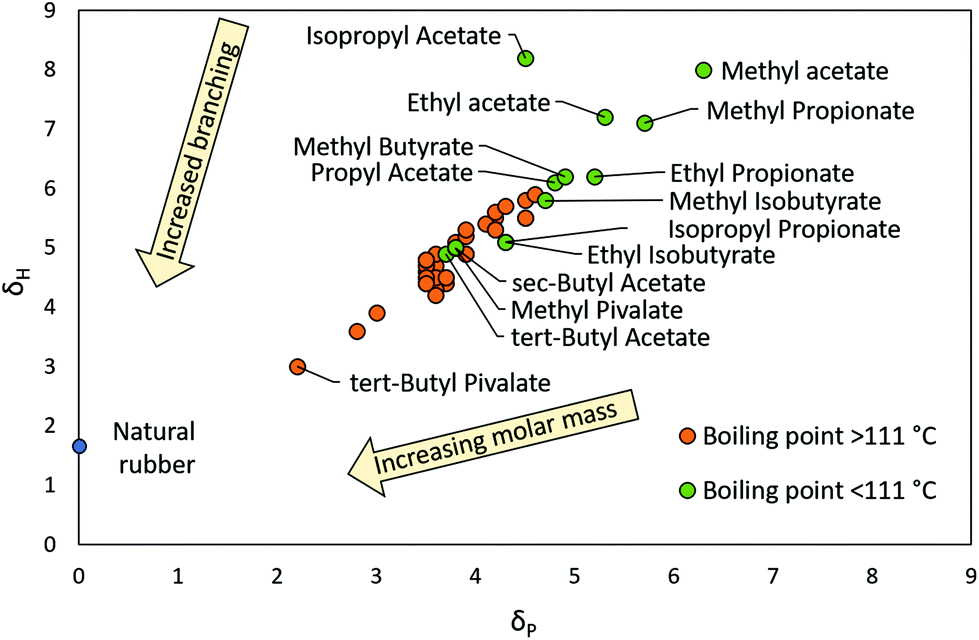
A methodical selection process for the development of ketones and esters as bio-based replacements for traditional hydrocarbon solvents - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC01132J

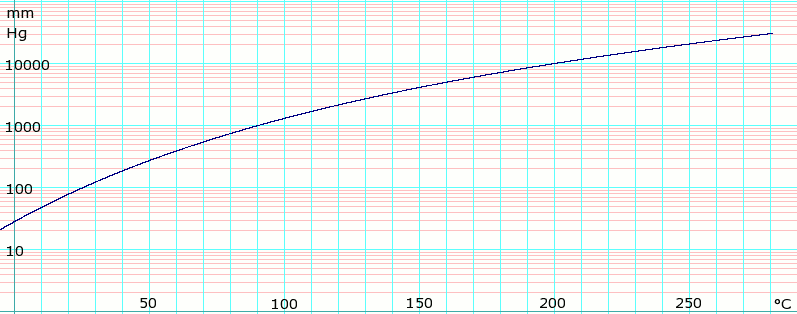
N-hexane vapor pressure curve and the chamber conditions at the start... | Download Scientific Diagram
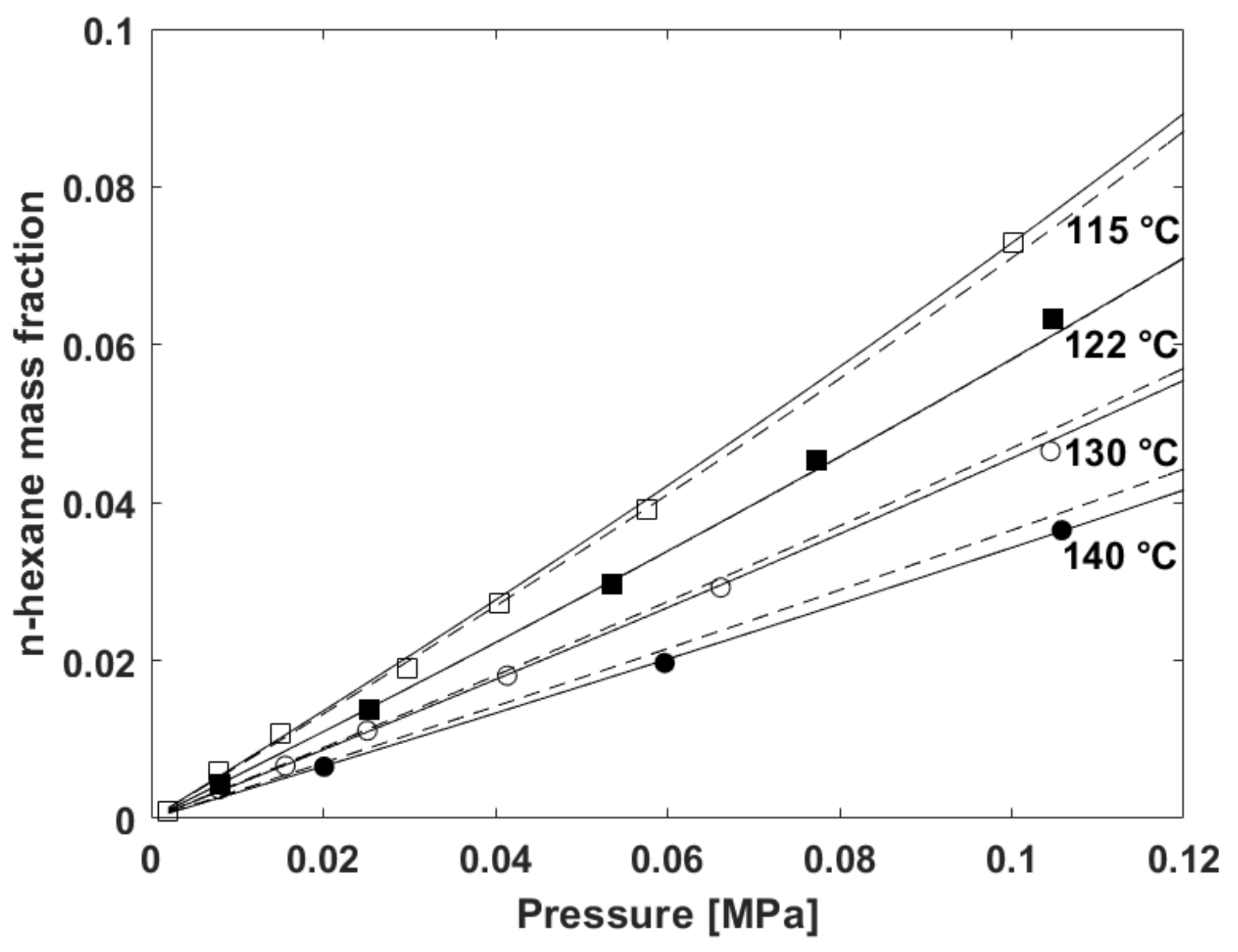
Polymers | Free Full-Text | Modelling Sorption Thermodynamics and Mass Transport of n-Hexane in a Propylene-Ethylene Elastomer | HTML
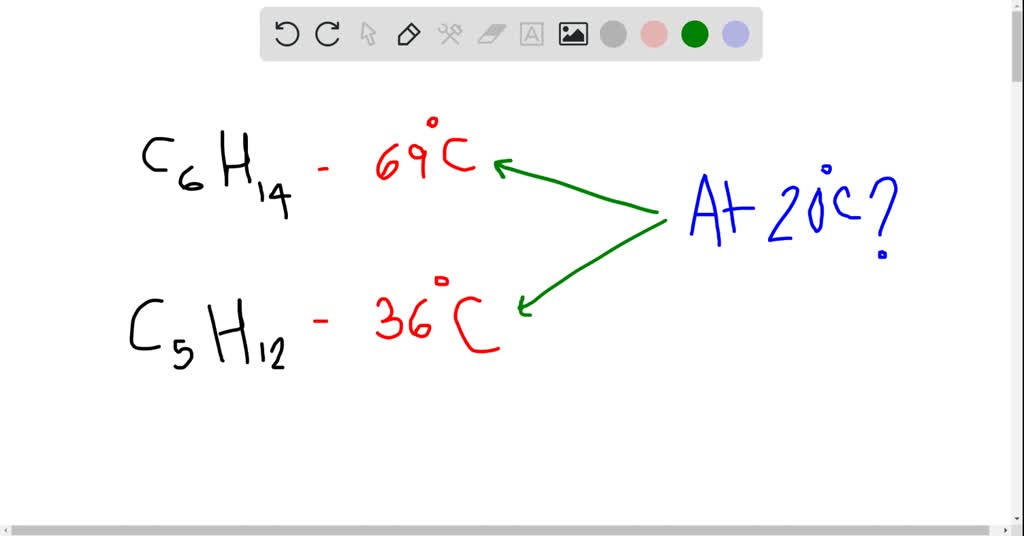
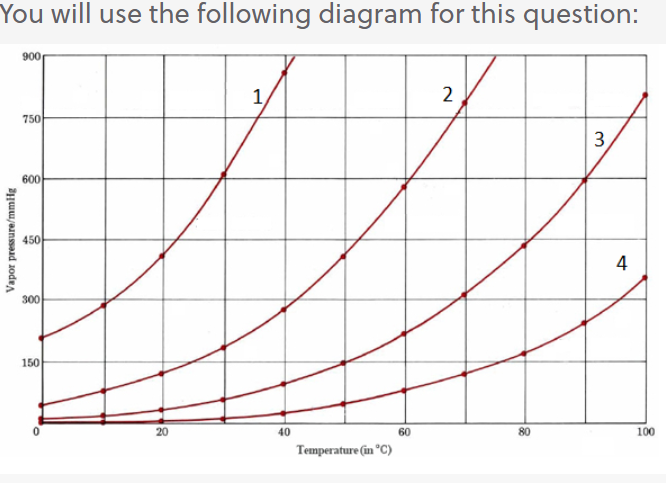
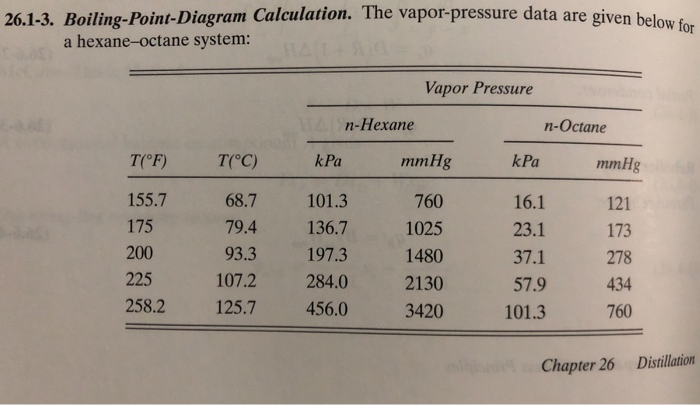
SOLVED:The normal boiling point of hexane, C_{6} H_{14}, is 69^{\circ} \mathrm{C}, and that of pentane, C_{5} H_{12}, is 36^{\circ} \mathrm{C}. Predict which of these compounds has a higher vapor pressure at 20^{\circ} \

Styrene Butadiene Rubber/Carbon Filler‐Based Vapor Sensors - Tabačiarová - 2016 - Macromolecular Chemistry and Physics - Wiley Online Library

SOLVED:The normal boiling point of hexane, C_{6} H_{14}, is 69^{\circ} \mathrm{C}, and that of pentane, C_{5} H_{12}, is 36^{\circ} \mathrm{C}. Predict which of these compounds has a higher vapor pressure at 20^{\circ} \