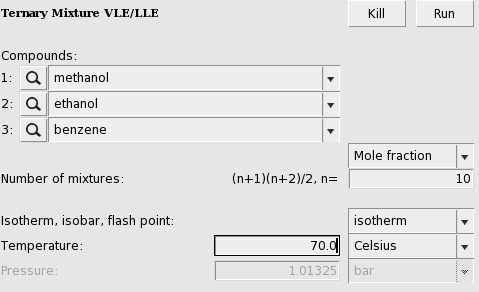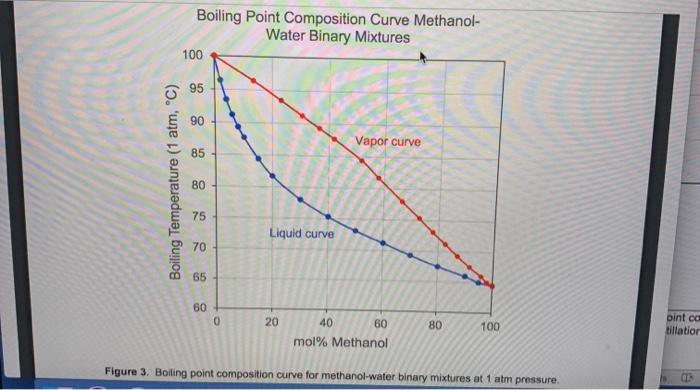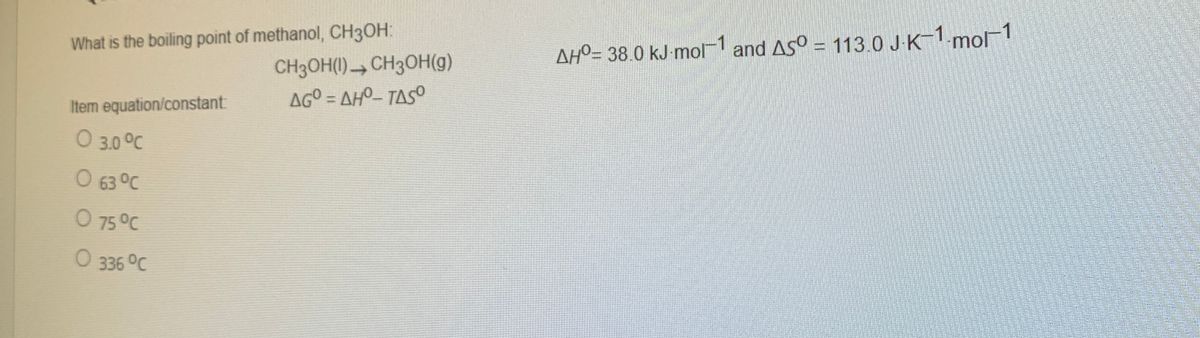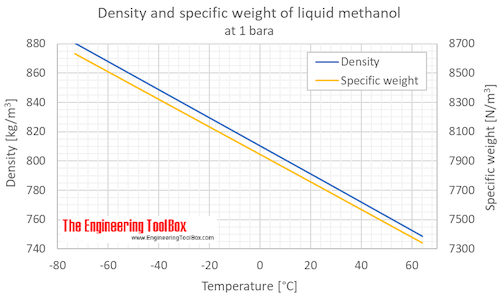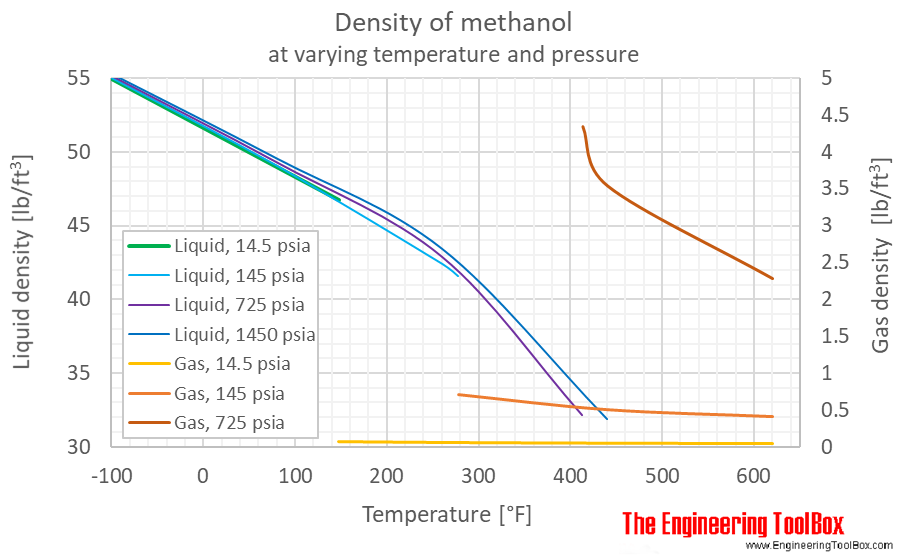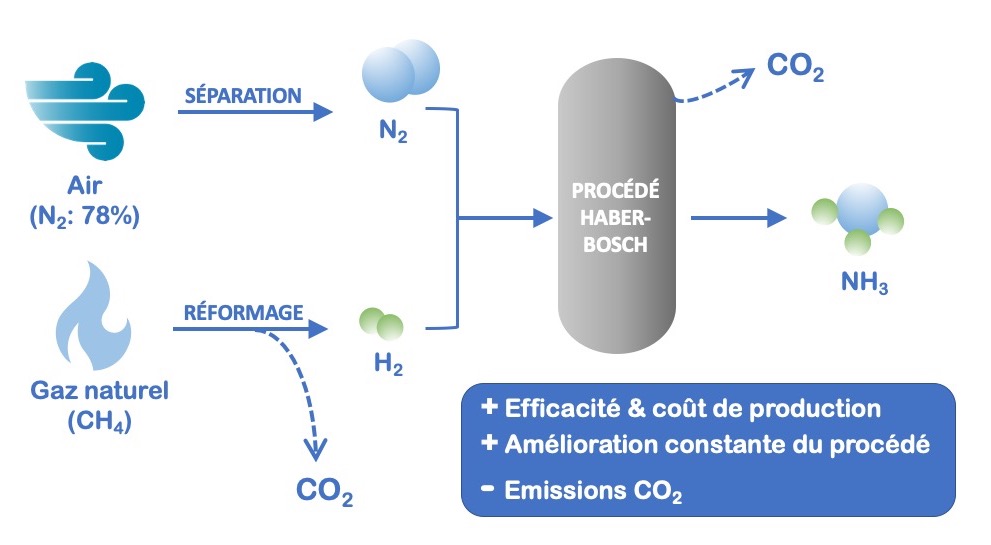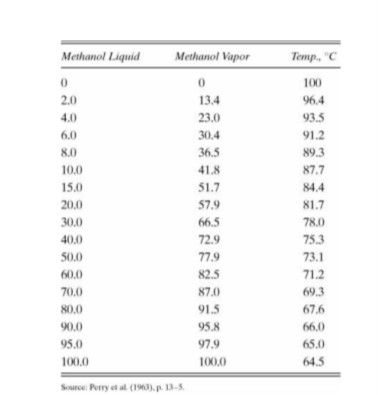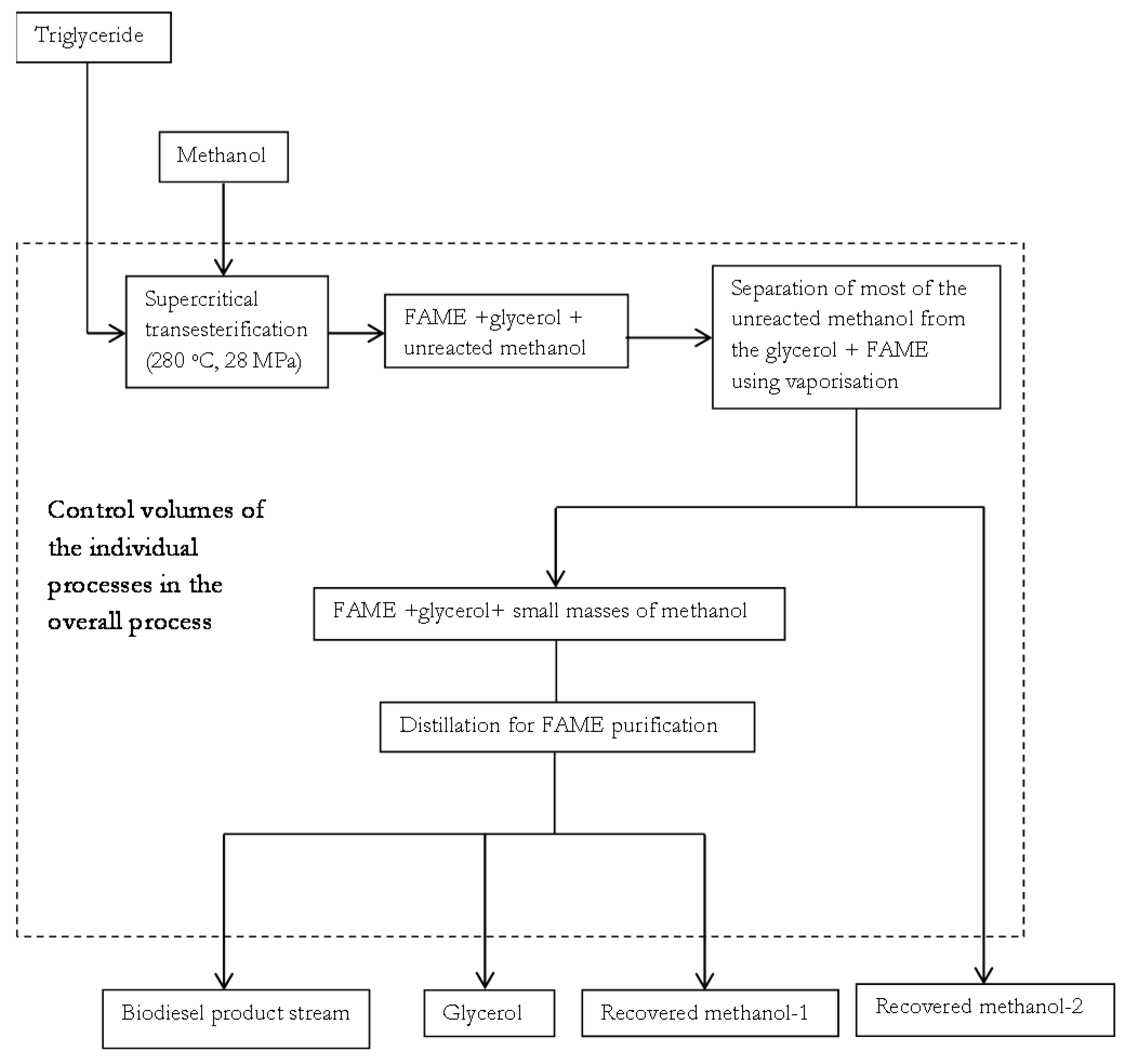
Sustainability | Free Full-Text | Catalyst-Free Biodiesel Production Methods: A Comparative Technical and Environmental Evaluation | HTML

Direct Utilization of Liquid Fuels in SOFC for Portable Applications: Challenges for the Selection of Alternative Anodes – topic of research paper in Chemical sciences. Download scholarly article PDF and read for

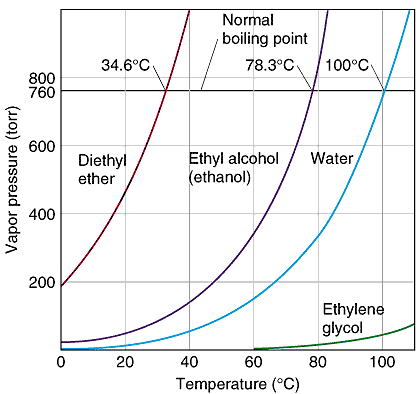
SOLVED:The boiling point of methanol is 65.0^{\circ} \mathrm{C} and the standard enthalpy of formation of methanol vapor is -210.2 \mathrm{~kJ} / \mathrm{mol} . Calculate the vapor pressure of methanol (in \mathrm{mmHg} )

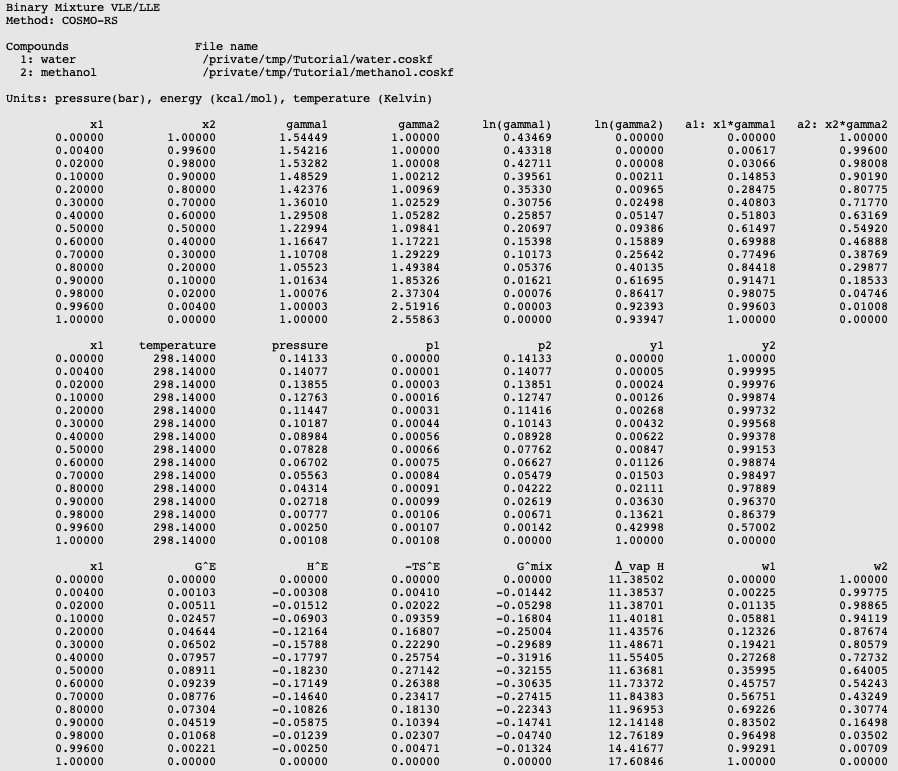
Influence of a Confined Methanol Solvent on the Reactivity of Active Sites in UiO‐66 - Caratelli - 2018 - ChemPhysChem - Wiley Online Library

Influence of a Confined Methanol Solvent on the Reactivity of Active Sites in UiO‐66 - Caratelli - 2018 - ChemPhysChem - Wiley Online Library
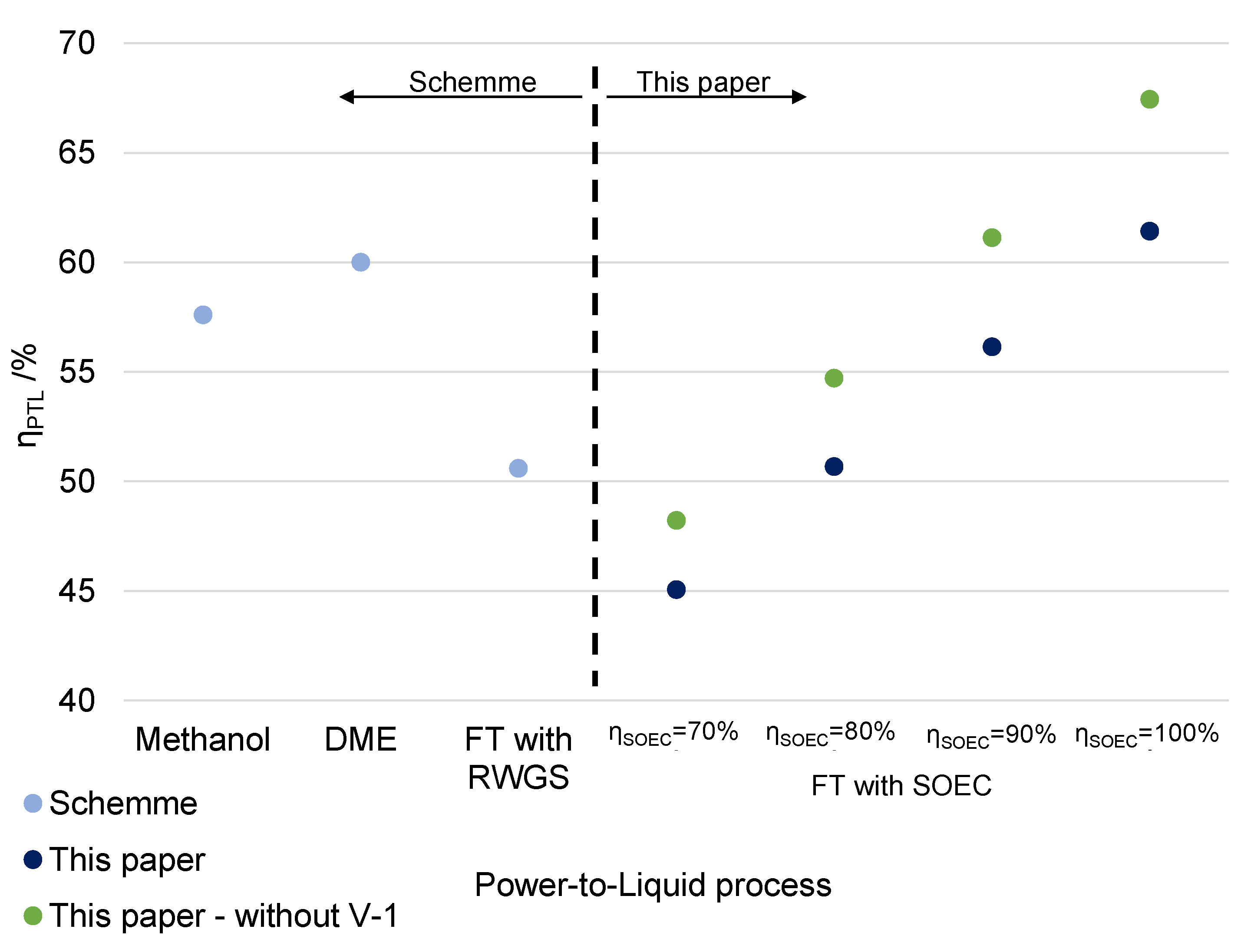
Processes | Free Full-Text | A Techno-Economic Assessment of Fischer–Tropsch Fuels Based on Syngas from Co-Electrolysis | HTML