![Isocyanide based [4+1] cycloaddition reactions: an indispensable tool in multi-component reactions (MCRs) - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC01562J Isocyanide based [4+1] cycloaddition reactions: an indispensable tool in multi-component reactions (MCRs) - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC01562J](https://pubs.rsc.org/image/article/2016/CC/c6cc01562j/c6cc01562j-s4_hi-res.gif)
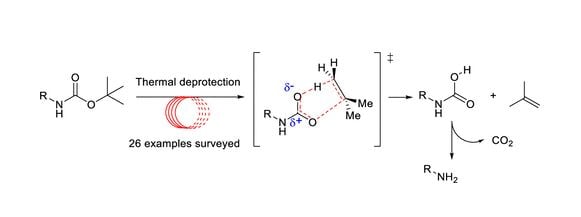
Isocyanide based [4+1] cycloaddition reactions: an indispensable tool in multi-component reactions (MCRs) - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC01562J

Progress in Lanthionine and Protected Lanthionine Synthesis - Denoël - 2018 - Chemistry – A European Journal - Wiley Online Library
Dual protection of amino functions involving Boc - RSC Advances (RSC Publishing) DOI:10.1039/C3RA42956C

PDF) p-Nitrobenzyloxycarbonyl (pNZ) as an Alternative to Fmoc for the Protection of Amines in Solid-Phase Peptide Synthesis

Advanced Organic Chemistry: Reaction Mechanism, Strategy, Applications.... - Advanced Organic Chemistry: Reaction Mechanism, Strategy, Applications.
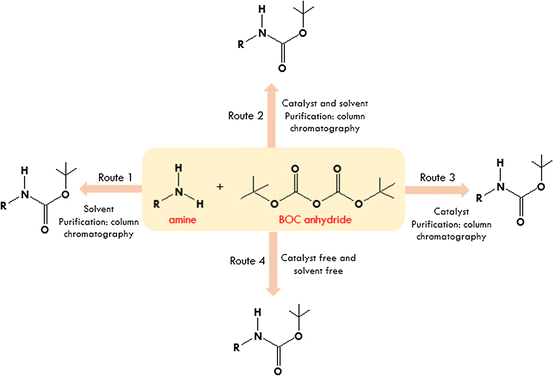
Efficient and expeditious chemoselective BOC protection of amines in catalyst and solvent-free media | SpringerLink

Deprotection of N‐tert‐Butoxycarbonyl (Boc) Protected Functionalized Heteroarenes via Addition–Elimination with 3‐Methoxypropylamine - Gulledge - 2020 - European Journal of Organic Chemistry - Wiley Online Library